|
The crucial atom in the hemoglobin protein is iron. Hemoglobin is a relatively large molecule, with a mass of about 65,000 u. Hemoglobin combines with oxygen and carbon dioxide, transporting these gases from one location to another in the body. The critical part of the red blood cell is a protein called hemoglobin. Without red blood cells, animal respiration as we know it would not exist. Red blood cells are cells that transport oxygen from the lungs to cells of the body and then transport carbon dioxide from the cells to the lungs. The chemistry of iron makes it a key component in the proper functioning of red blood cells. Transition metals have interesting chemical properties, partially because some of their electrons are in d subshells. Because iron has relatively massive atoms, it would appear even lower on a list organized in terms of percent by atoms rather than percent by mass. The first element appearing on the list that is not a main group element is iron, at 0.006 percentage by mass. Most of the elemental composition of the human body consists of main group elements. To Your Health: Transition Metals in the Body Each group is located in a different part of the periodic table. Elements are either metals, nonmetals, or semimetals. The valency of the compound is 1 and 1.\): Types of Elements.Some formulas through which we can make compounds by knowing the valency of their elements are as follows: Hydrogen Chloride Hence, the valency of vanadium are 2,3,4 and 5.įormulas of Compounds by Valence Electrons.Its electronic configuration is Ar 3d 3 4s 2.It is a transition element and most of these show variable valences.It will gain 3 more electrons to complete its octet.Its electronic configuration is 2, 8, 5.It shows two valences: 1 (or Cu(I)), known as cuprous, and 2 (or Cu(II)), known as Cupric.It is a transition element, and most of these elements have variable valences.The outermost shell is already filled, so it will not lose electrons.Sodium will lose 1 electron to complete its octet.Its electronic configuration is 2, 8, 1.Here are the formulas of Valence Electrons for some elements: Sodium Read More: Polytetrafluoroethene (Teflon) 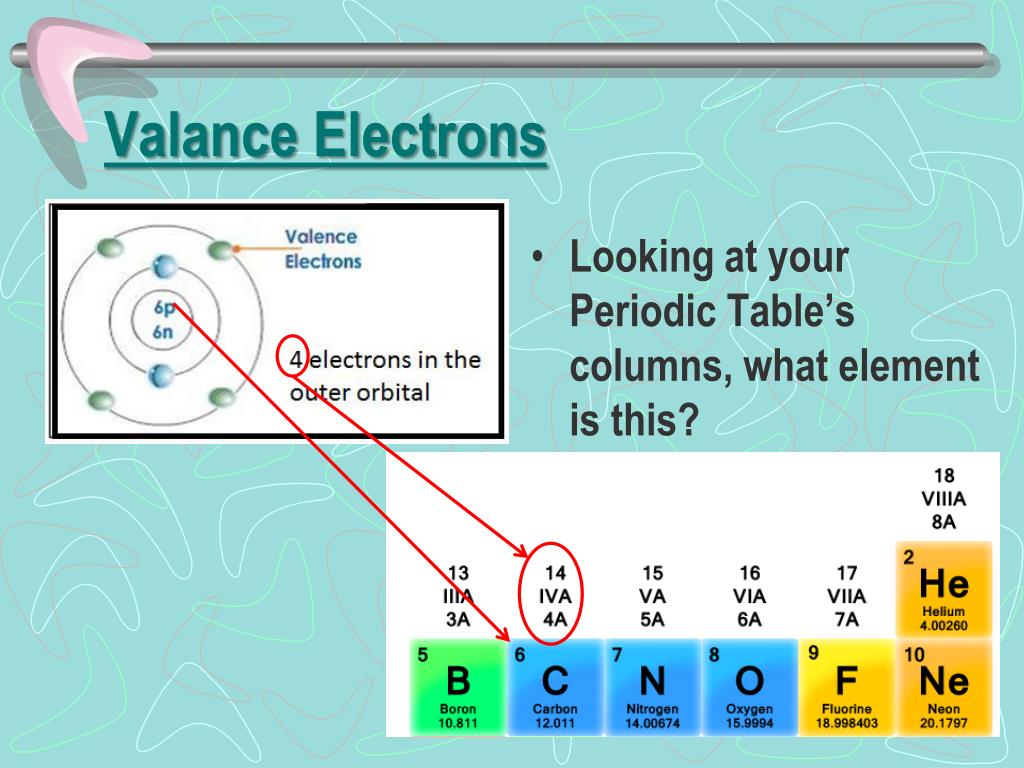 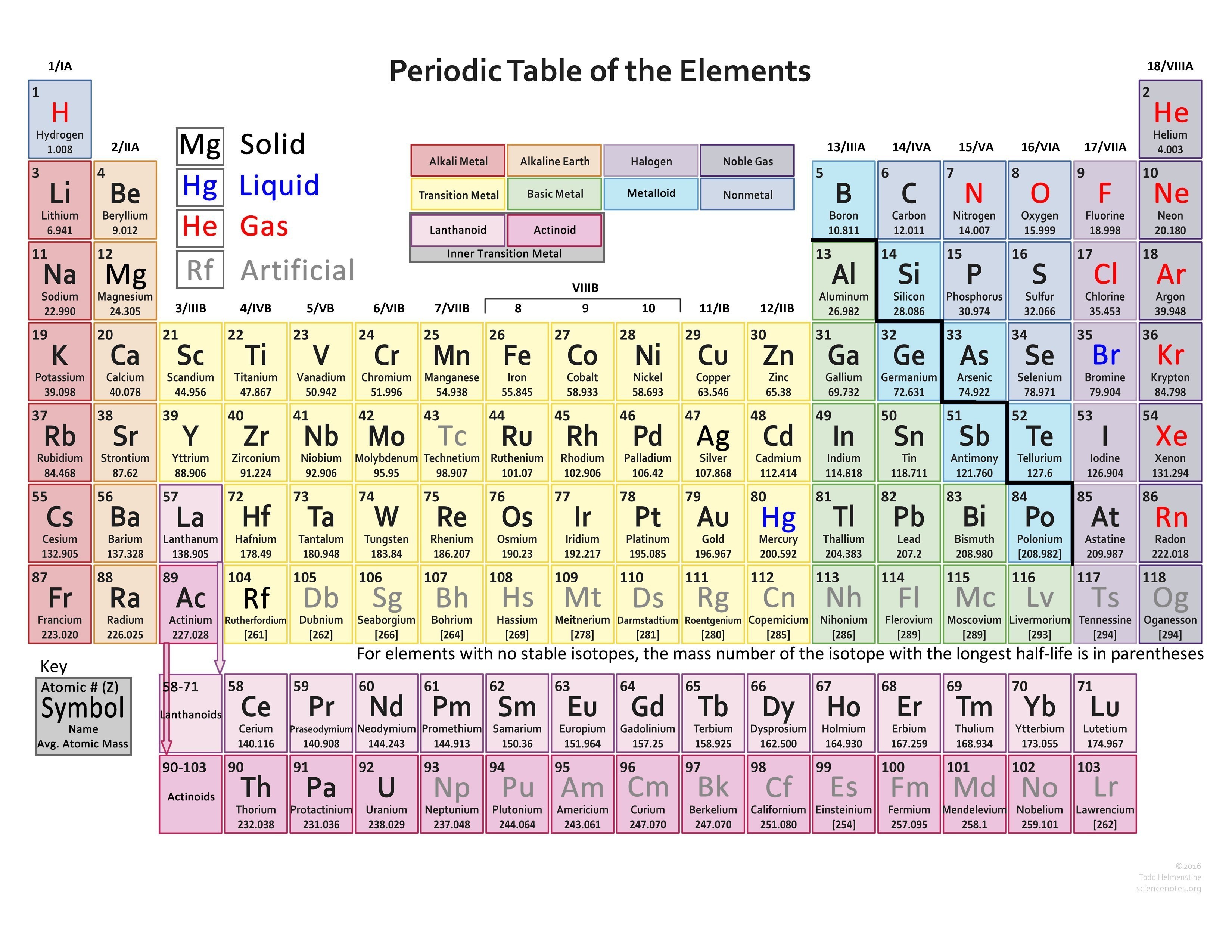
The periodic table of all the elements is mentioned below: If there are 8 electrons in the outermost shell, it means that the octet is completed already, and there is no need to lose or gain electrons.If there are 4 electrons in the outermost shell, it can lose or gain 4 electrons to complete its octet.If there are 5, 6, or 7 electrons in the outermost shell, it will gain some electrons to complete its octet.If there are 1, 2, or 3 electrons in the outermost shell, it will lose these electrons to complete its octet. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
